Theranostic Porphyrin–Lipid Nanoparticles (Por-NPs) for Atherosclerosis
Introduction
A new generation of “theranostic” nanoparticles has been shown to both detect and reduce plaques in the arteries . These porphyrin-lipid nanoparticles (Por-NPs) are designed to target atherosclerotic plaques and serve a dual purpose: imaging diseased sites and delivering therapy simultaneously. Atherosclerosis, the buildup of fatty inflamed plaques in artery walls, remains a leading cause of heart attacks and strokes . Traditional treatments like statins lower cholesterol but cannot directly visualize or eliminate existing high-risk plaques once they form . Por-NPs aim to fill this gap by identifying dangerous plaques and intervening to shrink them, breaking the vicious cycle of inflammation and plaque growth .

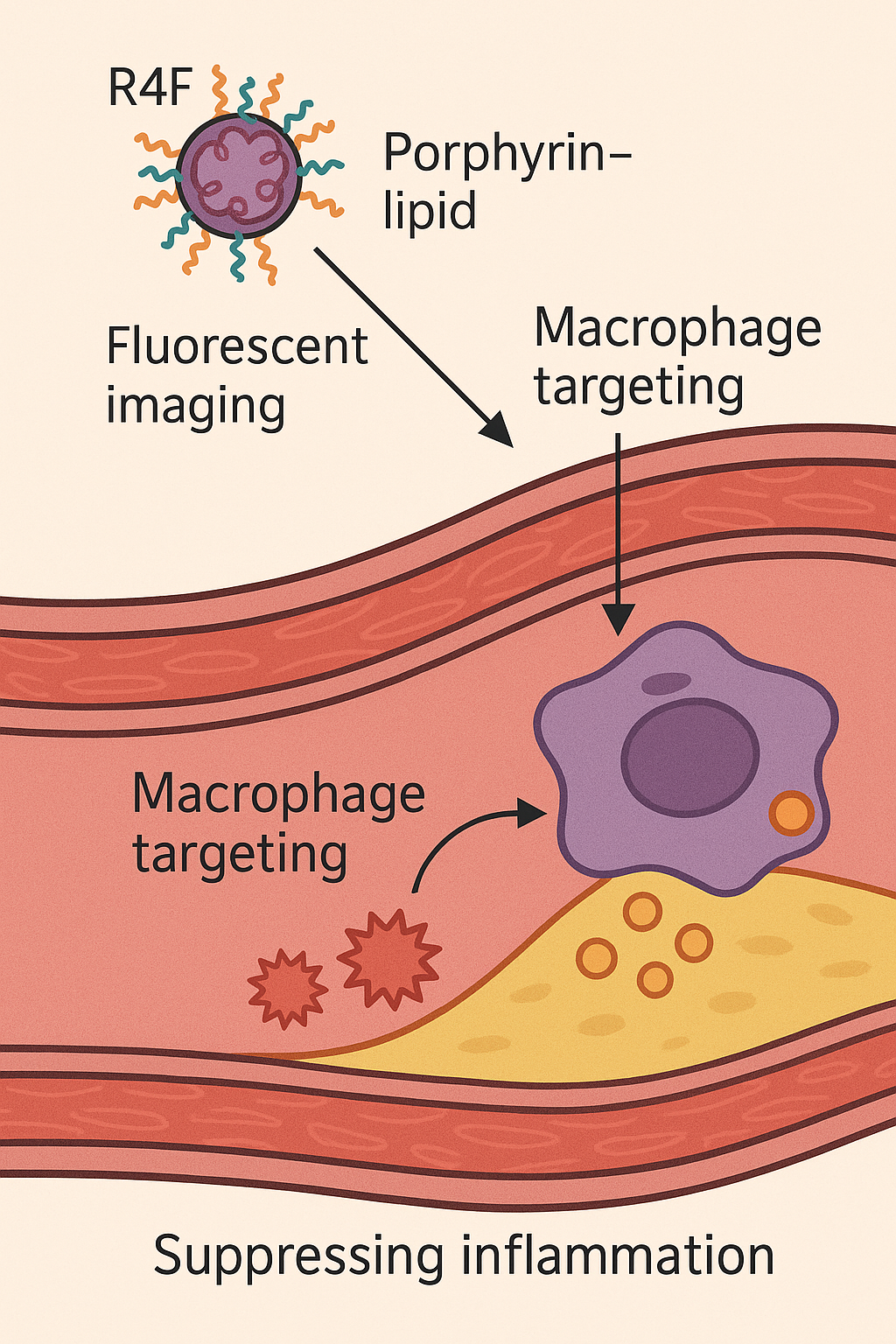
Illustration of how Porphyrin–lipid nanoparticles (Por-NPs) treat atherosclerotic cardiovascular disease by targeting plaque macrophages, promoting cholesterol removal, and suppressing inflammation (adapted from Nankivell et al.,
Materials Today Bio ). This theranostic nanoparticle platform combines a fluorescent, radiometal-chelating porphyrin-lipid core with an apolipoprotein-mimetic peptide shell. The unique design enables multimodal imaging of plaques (optical fluorescence and PET scanning) alongside biological therapy at the disease site.
Nanoparticle Design and Targeting Mechanism
Porphyrin-lipid nanoparticles are built to mimic key functions of high-density lipoprotein (HDL) and specifically seek out plaque macrophages. The nanoparticle’s core contains a porphyrin–lipid conjugate that naturally emits near-infrared fluorescence and can chelate Copper-64 for PET imaging . This allows the particles to be tracked in living tissue via both fluorescent imaging and positron emission tomography . The outer shell is decorated with a short peptide called R4F, an apolipoprotein A-I mimetic (also known as “reverse 4F”) that is designed to bind the scavenger receptor SR-BI on macrophages . By targeting SR-BI – a receptor abundantly expressed on plaque macrophages – the R4F coating directs the nanoparticles to atherosclerotic lesions for selective uptake . These Por-NPs are extremely small (~20 nm in diameter) , which is much smaller than a typical virus, facilitating deep penetration into plaques.
Once a Por-NP is taken up by a plaque macrophage, it effectively functions like a miniature HDL particle inside the cell. The R4F/SR-BI interaction and HDL-mimetic design help the foam cell offload excess cholesterol and lipids, preventing these macrophages from becoming overloaded “foam cells” . At the same time, the nanoparticle’s presence modulates the cell’s inflammatory pathways, helping to suppress the inflammatory response that drives plaque progression . Notably, researchers found that the anti-inflammatory effects of Por-NPs are partly independent of SR-BI and cholesterol efflux pathways, suggesting these particles may also neutralize pro-inflammatory signals directly .
Diagnostic Imaging Capabilities
Porphyrin-lipid NPs are intrinsically traceable by imaging, enabling early diagnosis of atherosclerosis and monitoring of treatment response. The porphyrin-based core fluoresces under infrared light and can be radiolabeled with ^64Cu, giving the particles a built-in dual imaging modality . In preclinical studies, infused Por-NPs were clearly visualized within plaques. PET scans detected the accumulation of ^64Cu-labeled Por-NPs in the hearts and arteries of atherosclerotic mice, and could even track the progression of plaque burden over time . Similarly, near-infrared fluorescence imaging revealed strong signals from Por-NPs co-localizing with CD68^+ macrophages inside plaque regions . This demonstrates that Por-NPs can serve as a contrast agent marking inflamed, macrophage-rich plaques using multiple imaging techniques. Importantly, the nanoparticles’ multimodal imaging capability could allow cardiologists to identify high-risk plaques earlier and more specifically than traditional scans .
Therapeutic Effects in Atherosclerosis Models
Beyond imaging, Por-NPs deliver tangible therapeutic benefits by altering plaque biology. In vitro experiments with macrophages showed that Por-NPs are readily internalized by these immune cells . Once inside, the particles significantly enhanced cholesterol efflux from the macrophages – by about 49% more than even natural reconstituted HDL particles . This means foam cells were able to unload a greater share of their cholesterol, which is a key step in stabilizing or regressing plaques.
Crucially, Por-NPs also damped inflammatory signaling in macrophages. Treated macrophages had dramatically lower levels of inflammatory cytokines: for example, mRNA for IL-1β was reduced by 88% and CCL5 (RANTES) by 75%, with corresponding protein levels of these cytokines dropping ~70–80% relative to controls . The nanoparticles suppressed activation of the NF-κB pathway and inflammasome components (e.g. NLRP3 by ~69%), which are central drivers of inflammation in atherosclerosis . Interestingly, follow-up tests revealed that these anti-inflammatory effects did not require SR-BI or cholesterol removal to occur . In other words, the Por-NPs inherently carry anti-inflammatory properties—likely by sequestering toxic lipid byproducts or directly interfering with inflammatory signaling—beyond just their HDL-like cholesterol transport function.
When tested in living animals, the Por-NPs demonstrated striking atheroprotective effects. Atherosclerosis-prone ApoE^-/- mice on a high-fat diet were treated with these nanoparticles in two disease models: one simulating early-stage plaque development and another modeling advanced, unstable plaques . In the early-stage disease group, mice receiving Por-NPs developed significantly smaller plaques (approximately 22% smaller by area) compared to untreated mice . In the advanced plaque model – which mimics vulnerable plaques prone to rupture – treated mice showed a remarkable 52% reduction in plaque size versus controls . These therapeutic effects on plaque burden were accompanied by a systemic anti-inflammatory impact: treated mice had 32% fewer circulating monocytes (inflammatory white blood cells) and an 81% reduction in monocyte/macrophage content within plaques relative to controls . Genes that drive inflammation in arterial walls were also “dialed down” in treated animals, indicating a healthier plaque environment .
Notably, the fate of the cholesterol removed from plaques appears to be benign. After doing their work in the arteries, the Por-NPs (and the cholesterol they carry) were mostly transported to the liver for disposal, just as natural HDL would do . Researchers observed no harmful accumulation in the liver or other organs, suggesting the nanoparticles safely integrate into the body’s normal cholesterol metabolism and clearance routes . As Dr. Victoria Nankivell, the study’s lead author, summarized: “These nanoparticles don’t just detect arterial plaque… they can also suck it up and take it to the liver, lowering inflammation” .
Future Directions and Clinical Translation
The promising results with Por-NPs so far have been achieved in preclinical (in vivo) models of atherosclerosis – primarily mouse models of arterial plaque disease . The ability to simultaneously image and treat plaques marks an exciting step toward so-called “theranostic” cardiology. According to the investigators, Porphyrin-lipid NPs represent a potential nanoscale theranostic platform for atherosclerotic cardiovascular disease . In other words, a single agent can play both diagnostic and therapeutic roles, as demonstrated by its multimodal plaque imaging and atheroprotective effects in animals .
Having shown efficacy in animals, the next challenge is to translate this nanoparticle technology into clinical trials. The research team is now exploring further development of Por-NPs for clinical use, with the goal of complementing existing heart disease treatments and improving patient outcomes . Many steps remain, including scaling up nanoparticle production, rigorous safety evaluation, and regulatory approval before human studies can begin. Nonetheless, the concept holds great potential. In the future, Por-NPs (or similar HDL-mimetic nanomedicines) could be used alongside standard therapies to not only visualize dangerous plaques non-invasively but also actively reduce plaque size and inflammation before they cause heart attacks or strokes . This approach exemplifies the growing field of nanomedicine, where tiny engineered particles are employed to both see and treat disease processes at the same time .
Key Points
- Diagnostic capability: Yes. Porphyrin-lipid NPs can be tracked by near-infrared fluorescence and PET imaging, enabling identification of atherosclerotic plaques in vivo .
- Therapeutic capability: Yes. The R4F-coated NPs actively reduce plaque burden and inflammation by targeting cholesterol-laden macrophages and modulating their biology .
- Dual function (Theranostic): Both. These nanoparticles serve as a combined diagnostic and therapeutic tool – one platform for imaging plaques and treating them simultaneously .
- Stage of development: Tested in vivo in animal models (mice), showing significant efficacy . Now under development towards clinical trials, with hopes to translate this approach into human heart disease management.
Discover more from RETHINK! SEEK THE BRIGHT SIDE
Subscribe to get the latest posts sent to your email.