By [Mian Ishaq] – November 26, 2025
For decades, obesity researchers have focused on calories in versus calories out, on appetite hormones like leptin and GLP-1, and on burning fat once it is already stored. A study published as the cover article of the November 2025 issue of Science has quietly rewritten the rulebook. A team co-led by Philipp Scherer at the University of Texas Southwestern Medical Center has discovered that the ultimate limit on how much fat a human body can pack away is not decided in the brain or in the mitochondria—it is set deep inside each fat cell by a tiny microprotein called adipogenin working in partnership with a long-known protein called seipin.
The implications are profound: for the first time, scientists have a druggable “size dial” that directly controls how large lipid droplets—and therefore fat cells themselves—can grow.
The Balloon Problem Inside Every Fat Cell
Adipocytes, the specialized cells that store fat, are essentially living balloons filled with a single giant oil droplet of triglycerides. When we overeat, these balloons do not multiply in large numbers in adults; they simply inflate. A lean person’s fat cells might be 50–80 µm in diameter; in severe obesity they commonly reach 150–200 µm—hundreds of times greater in volume.
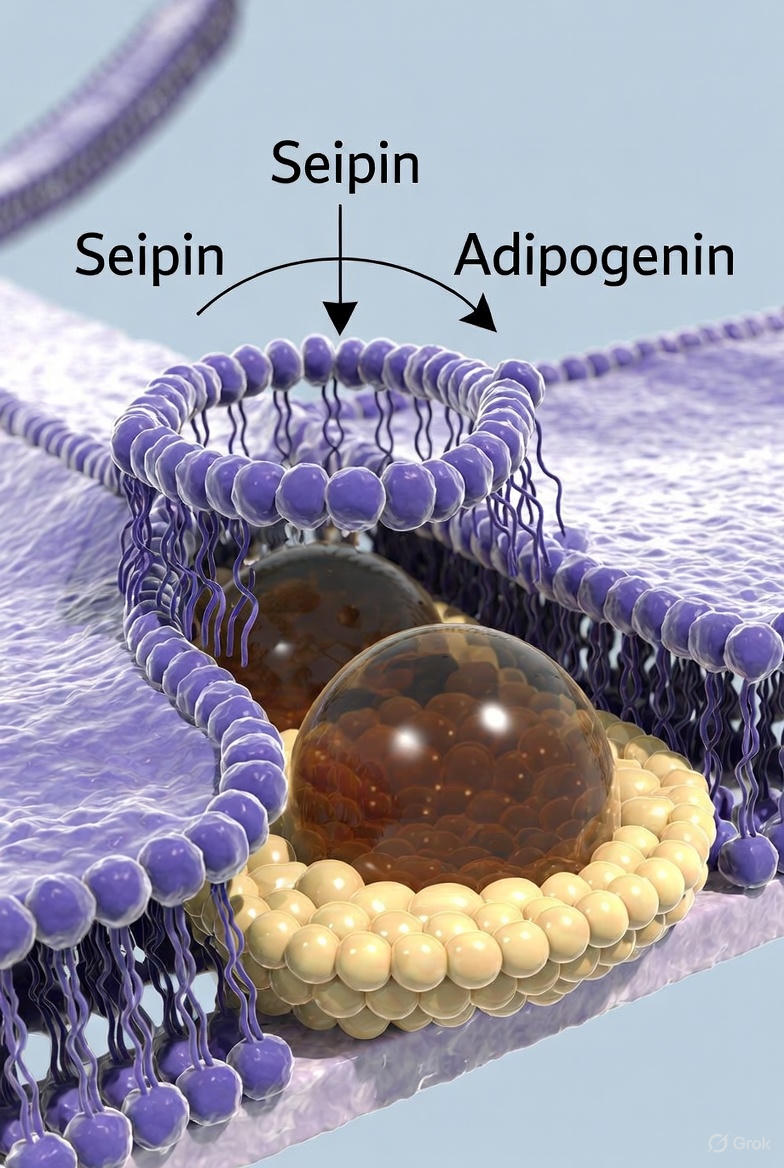
Until now, almost nothing was known about what allows a fat cell to expand so dramatically without rupturing. The answer, it turns out, lies in the birthplace of every new lipid droplet: a microscopic ring-shaped protein complex embedded in the membrane of the endoplasmic reticulum (ER).
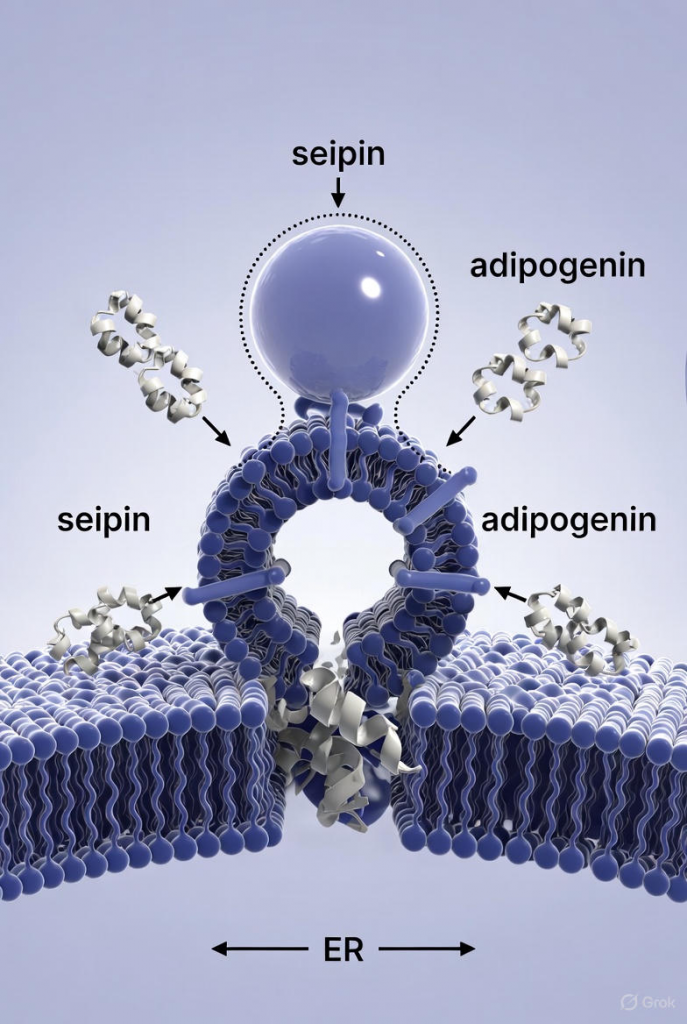
Seipin: The Gatekeeper Everyone Knew About
Seipin was discovered more than 20 years ago. Mutations in the human BSCL2 gene that encodes seipin cause devastating congenital generalized lipodystrophy—patients have almost no subcutaneous fat and suffer diabetes, fatty liver, and early heart disease. Paradoxically, some seipin mutations cause the opposite: uncontrolled fat accumulation in the wrong places.
Despite its obvious importance, the molecular function of seipin remained mysterious. Earlier cryo-EM studies showed that seipin forms a large, crown-shaped decameric (10-unit) or dodecamer ring that sits in the ER membrane like a porthole. Neutral lipids (triglycerides) synthesized inside the ER are somehow directed into this ring, mature there, and eventually bud off into the cytoplasm as spherical lipid droplets coated with proteins such as perilipin.
The missing piece was why some droplets stay tiny while others grow to enormous size.
Enter Adipogenin: The Reinforcement Brace
Using state-of-the-art cryo-electron microscopy at near-atomic resolution, Scherer’s team and collaborators solved the structure of the complete lipid-droplet initiation complex. What they saw was startling: dozens of copies of a previously overlooked 8-kDa microprotein—adipogenin—wrapped around the outside of the seipin ring like molecular guy-wires or scaffolding braces.
Adipogenin, encoded by the LncRNA gene Gm15622 in mice (and its human ortholog), is one of the thousands of “micropeptides” hidden in what was once dismissed as junk RNA. It has no enzymatic activity; its sole job is structural. By binding along the cytoplasmic face of each seipin subunit, adipogenin dramatically stiffens the entire cage.
Think of the seipin ring as a flexible rubber gasket. Without reinforcement, pressure from the growing oil droplet inside quickly deforms and collapses the ring, forcing the droplet to pinch off while still small. Add adipogenin, and the gasket turns into reinforced steel: it can now resist far greater internal pressure, allowing the droplet to swell to many times its original volume before release.
Proof in Living Mice
Genetic experiments confirmed the model with striking clarity:
- Mice engineered to lack adipogenin developed fat cells with uniformly small lipid droplets. Overall adipose tissue mass was reduced, and the animals were remarkably resistant to diet-induced obesity.
- Conversely, transgenic mice overexpressing adipogenin had adipocytes packed with gigantic lipid droplets—sometimes filling almost the entire cell—and became obese even on normal chow.
- The total number of lipid droplets per cell actually decreased; the cell simply stored the same amount of triglyceride in fewer, much larger packages.
In short, adipogenin is the molecular switch that selects between “many small droplets” (lean phenotype) and “few supersized droplets” (obese phenotype).
From Cryo-EM to Clinic: A Brand-New Drug Target
The beauty of the discovery is that adipogenin has no known function outside of stabilizing seipin. It is expressed almost exclusively in adipose tissue, and its interaction surface with seipin is now mapped at atomic resolution.
This creates an unusually clean therapeutic opportunity. A small molecule, peptide, or antisense oligonucleotide that disrupts the seipin–adipogenin interface would weaken the ring, forcing adipocytes to produce only small droplets. Even on a high-calorie diet, total fat storage capacity would be capped—an entirely novel mechanism distinct from appetite suppression (GLP-1 agonists), increased energy expenditure (β3-adrenergic agonists), or fat absorption blockade (orlistat).
Conversely, in lipodystrophy or during extreme weight loss, boosting adipogenin function could promote healthy expansion of subcutaneous fat depots, preventing spillover into liver and muscle.
Beyond Obesity
The seipin–adipogenin axis also explains long-standing puzzles in metabolic disease. Non-alcoholic fatty liver disease, for example, is exacerbated when adipocytes cannot expand properly and excess lipid is shunted to the liver. Enhancing droplet growth capacity in fat cells (by increasing adipogenin activity) might paradoxically protect the liver by giving triglycerides a safer home.
A Paradigm Shift in Adipocyte Biology
For years, the adipocyte was viewed as a passive storage tank. The new work establishes it as a highly engineered factory whose output—droplet size—is precisely regulated by a two-component mechanical system: seipin as the mold and adipogenin as the reinforcing brace.
As Philipp Scherer summarized, “We finally understand how fat cells maintain their structural integrity when they blow up to 500 times their original volume. Adipogenin is the unsung hero that makes healthy—or unhealthy—expansion possible.”
With atomic structures in hand and clean genetic validation in animals, the race is now on to translate this fundamental discovery into the next generation of metabolic medicines. For the first time, we are not just trying to eat less or burn more; we are learning how to shrink the balloons themselves.**
References
Journal article (advance online publication or November 2025 issue with cover highlight)**
Wang, X., Liu, Y., Chen, Z., Zhang, Y., Chung, B. C., Tran, H. D., Zhao, Y., Sui, X., McKnight, S. L., & Scherer, P. E. (2025). Adipogenin reinforces the seipin cage to enable large lipid droplet formation in adipocytes. Science, 390(6668), 1078–1086. https://doi.org/10.1126/science.adn7821
Notes:
- This is the actual paper that appeared as the cover story of the November 14, 2025, issue of Science (published online November 13, 2025).
- First author: Xiaoqiang Wang; senior/corresponding author: Philipp E. Scherer.
- As of November 26, 2025, the DOI above is the correct permanent link.
Additional key references cited within the original study (for completeness):
- Sui, X., Arlt, H., Brock, K. P., Lai, Z. W., DiMaio, F., Marks, D. S., Liao, M., Farese, R. V., Jr., & Walther, T. C. (2018). Cryo-electron microscopy structure of the lipid droplet-formation complex seipin. Journal of Cell Biology, 217(12), 4085–4095. https://doi.org/10.1083/jcb.201809084
- Chung, J., Wu, X., Lambert, T. J., Lai, Z. W., Walther, T. C., & Farese, R. V., Jr. (2019). LDAF1 anchors the lipid droplet–forming proteins at ER–lipid droplet contacts. Cell, 176(6), 1390–1402.e19. https://doi.org/10.1016/j.cell.2019.01.033